Evaluating Nadofaragene Firadenovec Phase III Clinical Trial Results as a New Gold Standard for BCG-Unresponsive Bladder Cancer - Trinity Bivalacqua & Nicholas James
January 25, 2021
that this therapy will become a standard of care in the treatment of BCG unresponsive disease.
Biographies:
Trinity J. Bivalacqua, MD, Ph.D., R. Christian B. Evensen Professor of Urology and Oncology, Director of Urologic Oncology at the James Buchanan Brady Urologic Institute, Johns Hopkins Medicine, Baltimore, MD
Professor Nicholas James, MBBS, FRCP, FRCR, Ph.D., Professor of Clinical Oncology at the Institute of Cancer Research at Royal Marsden Hospital, London
Ashish Kamat, MD, MBBS, President, International Bladder Cancer Group (IBCG), Professor of Urology & Cancer Research, MD Anderson Cancer Center, Houston, Texas
Nadofaragene Firadenovec for BCG Unresponsive NMIBC - Girish Kulkarni
Results of the SUO CTC Phase III Nadofaragene Firadenovec Trial for BCG Unresponsive NMIBC - Colin Dinney
Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial.
Ashish Kamat: Welcome to UroToday's Bladder Cancer Center of Excellence. I'm Ashish Kamat from Houston and it's my pleasure to welcome today, Dr. Trinity Bivalacqua from Baltimore John Hopkins Medicine and Professor Nick James from the UK.
Today we're going to talk about a recent development as far as a publication is concerned on the impact of nadofaragene firadenovec, which is the adenovirus interferon gene therapy Phase III clinical trial, which was published in The Lancet Oncology. Trinity is going to take the pro side as to the question of whether this is a major advancement for our patients with BCG unresponsive non-muscle invasive bladder cancer. And then Nick is going to take the opposing side and question Trinity and me as well as to why we think this is actually a major advance based on the data. With that Trinity, the stage is yours.
Trinity Bivalacqua: Thanks, Ashish. My job today is to provide you with the results of the Phase III clinical trial. And then I will do my best to express my thoughts on why this will be a mainstay for treatment of BCG unresponsive disease.
These are my disclosures. I am a consultant for FerGene, who is the sponsor of the Phase III trial.
Here are the authors and I will present as a member of the team that conducted this trial through the SUO CTC, which looked at intravesical instillation of nadofaragene firadenovec, which I will call adenoviral gene therapy or Ad-interferon in BCG unresponsive disease.
Here, you can see the patient population that we studied. This was a single-arm open-label study evaluating Ad-interferon in high-grade BCG unresponsive non-muscle invasive bladder cancer by the definition proposed by the FDA in 2018. The cohort enrolled 157 patients, both CIS plus or minus papillary disease or pure papillary Ta or T1 disease. A high percentage of patients had CIS, as we will describe. The treatment regimen was intravesical Ad-interferon. The dose you see here, every three months for a total of one year with the option of 24 months of maintenance therapy. Our endpoints were recurrence-free survival as you can see in the study.
I'd like to point out that there was a mandatory five-point biopsy for all patients that were enrolled in the trial at 12 months. Patients that had no signs of a recurrence seen by cystoscopy or by cytology at 12 months were mandated to undergo biopsies of the mucosa in targeted biopsies or in the posterior right and left sidewalls trigone and dome.
Here are the baseline characteristics. The majority of the patients had CIS plus or minus papillary disease. I'd like to point out that the majority of the patients that had CIS had greater than three intravesical installations of BCG and patients with high-grade T1 or Ta disease, also 30% of more had greater than three BCG instillations. So this was a heavily treated population.
Here are the results. Looking at high-grade recurrence-free survival at three months in patients with CIS plus or minus papillary disease, 53% of the patients had no signs of disease. And at 12 months, 24%. if you look at the cohort that did not have CIS, this number was higher at three months, 72% and at 12 months, 43%.
If we look at high-grade recurrence-free survival in patients who achieved a CR at three months and this is how you're seeing a lot of these data from the newer trials that are being presented both at the AUA, at ASCO, ESMO this is how they're being presented. Once again, patients that achieved the CR at three months, at 12 months, patients with CIS and a 45% disease-free survival. And those with papillary disease had 60%. You can see that the median duration of CR was about 10 months in CIS patients and over one year for those with papillary disease.
If you look at patients that were found to have CIS at the 12 month mandated biopsy, actually five of those patients would not have been considered presence of disease. Based on clinical features alone, a response rate at 12 months would have been reported in the CIS cohort of 27% and those with papillary disease, 48%.
If we look at disease recurrence and progression by 12 months, think it's important to note that eight patients in the entire cohort did have progression to muscle-invasive disease. However, this is not unexpected considering that this was a heavily pretreated population with multiple high-risk features, including the majority of the patients having CIS and over a third had a history of T1 disease. And importantly, there were no deaths from bladder cancer.
26% of the entire cohort did undergo a cystectomy and so this was a total of 40 patients. And you can see here that the median time to cystectomy in patients with CIS was about nine months. And those with papillary disease was about eight months.
We look at adverse events. This is one of the important points about this treatment, this intravesical treatment. There were very few serious adverse events and with only about three or 2% considered to be related to the drug that were grade 3s.
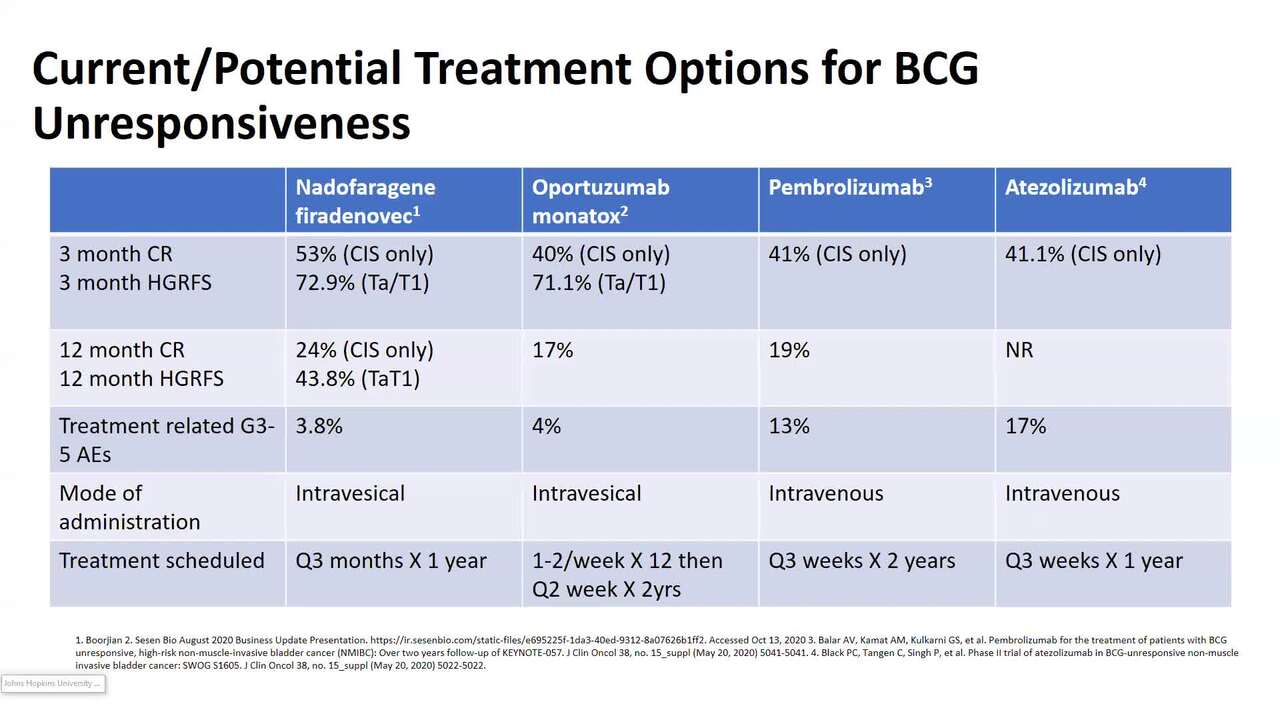
Why is this a major advance in the field of non-muscle invasive bladder cancer? Well, I think the best way to describe this would be to look at the current or potential treatment options for BCG unresponsive disease. These are either comparing pembrolizumab, which is already FDA approved for BCG unresponsive disease, and then compared to nadofaragene or Ad-interferon, We're looking at atezolizumab which was the Cooperative Group trial that actually was halted as well as our other, vicinium, which is currently under approval at the FDA. I'd like to point out that Ad-interferon is currently being looked at by the FDA and has not been approved.
If we look at the three-month complete response rate, Ad-interferon was superior to all of the other agents that are either approved or being investigated currently. If we look at 12 month CR rates, once again, superior to all those that are either approved or being studied. And I think one of the important points here is, is that there was very little grade 3 to 5 adverse events; 3.8% for Ad-interferon compared to the systemic agents, which is pembro and it is atezolizumab. The other thing that makes this such an important advance is the ease of installation, which is intravesical and every three months. The dosing regimen is every three months compared to that of vicinium, which is one to two times per week and every other week for upwards of two years. This makes it much easier for patients to tolerate. And when you look at compared to intravenous installation, which is every three weeks, at least in the trials that were looked at these two systemic agents.
The other thing that I think is important to note is that with Ad-interferon, it had such good response rates in papillary disease. It allows us to be able to expand this to look at intermediate-risk disease, for which currently a Phase III trial is being proposed. And hopefully, we'll look at its utility in intermediate low-grade papillary disease.
Looking to the future. I will acknowledge that we need to do better than the current data that we have for all agents and as well as Ad-interferon. And I think what you're seeing right now in all the trials that are being investigated is the use of combination therapy, either intravesical with a systemic agent. And I think that ultimately we need to determine a good base intravesical therapy, an agent to be used as intravesical therapy, to combine with systemic treatment in BCG unresponsive disease. And I'm also interested in seeing the role of radiation therapy with intravesical therapy. Currently, there are trials that are looking at radiation combined with systemic immune checkpoint in BCG unresponsive disease. And I think for me, at least from looking at that early data, it would make more sense to be combined with intravesical chemotherapy in BCG unresponsive disease.
That's all I have for now. I'm happy to answer additional questions once we hear from Professor James.
Ashish Kamat: Thanks, Trinity. And if you can just put up your data slide, the slide that had the table on there, that'd be great. And I'm going to take a back seat here a little bit and let Nick be the questioner, I guess, in this case, and put you on the spot a little bit and ask you and reveal to our audience why he thinks this might not be as much of an advance as you and I, and a lot of the urologists on the publication and in the field, think it is. Nick, the stage is yours.
Nicholas James: Thank you. Thank you very much. I have to say, this is a fantastic paper and it's a fantastic piece of work. I feel a bit uncomfortable arguing that it's not an advance because it clearly is an advance. It's a novel therapeutic modality. I've done adeno trials myself in prostate cancer. I know how hard these things are to set up, let alone do a Phase III trial. I think the authors and the whole team had to be congratulated on doing this. But there is a but. I think there's a number of things I'd criticize this for.
First, who on earth thought of that name? Trinity, you've got about five variants of it as you went through and you ended up on Ad-interferon which is about the only one that kind of makes sense. But leaving that aside, I'm very uncomfortable the trial design on a number of grounds. Firstly, I do like randomized trials and I know the FDA has accepted in this setting there's no suitable comparator, but then you've put a whole load of comparators that you could have had. And to which I would also have added cystectomy and hypothermic mitomycin as potential comparators as well. I think not saying a randomized trial is a bit of a cop-out given that there are things that are approved in this setting. Why are you not randomizing against other approved therapies or potentially approved therapies? I think that is an issue.
The second problem I have for the endpoint is if you take a sort of take a step back and think what's the important thing from the patient's point of view? They don't want to be coming back for a cystectomy later when they should have had the cystectomy straight up with a lot of anxiety in between. Potentially, always the risk of progression and you did show there was progression to muscle invasion. They just didn't have long enough to die, did they? We know that half of patients who get muscle-invasive bladder cancer are going to die a bit roughly eventually. You've got a window of opportunity to not die of bladder cancer by taking the bladder out now, which you potentially lose if you progress. I think, having a median response of eight or nine months and then you have your bladder out anyway, kind of what is the point really? And I would argue the same for all of the other treatments you've got here, the pembro, atezo, and so on. Is that if you're going to fail in nine months' time, what is the point of spending 30,000 pounds to defer that?
The second thing was that clearly is this lumps in... Or third or fourth thing, I'm not sure what number I've got to... Lumps in T1, G3 disease, as it tends to get called here and CIS and they clearly are not the same disease. They've got different underlying biology. They clearly behave differently in the trial and they behave differently if you pick the trials that have been done with hyperthermic mitomycin and with radiotherapy in this setting, different effects were seen for the treatment modalities in those two different disease settings. Lumping them in, I think is potentially an error as well. They should be separated.
The next thing I would be concerned about and we're coming back to the discussion we were having about COVID vaccines is the adenovirus itself, whether it will be immunogenic. I would question how many times you can repeat the dose before you start mounting a mucosal response to the vector and nullify any effect from the payload in the vector.
I think for all of those reasons, I think this is a very interesting proof of concept. I don't think any of these things are standard of care. They're not used in the UK and I don't think they should be. I think if you're just postponing cystectomy by a few months, even by a year, I don't think that's a good enough outcome from the point of view of the patients. As I said, I think it's a fantastic piece of work and I think doing a randomized trial earlier against things that are already working, but not very well like BCG, I think is really very interesting. I think if this outperforms BCG, fantastic, but I think this setting, I'm not convinced it's a standard of care. I'm not sure anything listed as a standard of care, to be honest.
Ashish Kamat: Let me jump in a little bit, Trinity, while you're responding to Nick's comments and address one question, which I hear coming up very often, which is why was a randomized trial and study not done? And that goes back to essentially a lot of discussions that we as a collective have had with the FDA about doing single-arm studies in this BCG unresponsive space, simply because as has been shown by many failed attempts, patients are not willing to be randomized to radical cystectomy.
And at the time, even today, there is no standard of care other than pembrolizumab that is effective in this patient cohort. Based on a strict definition of BCG unresponsive disease that the international bladder cancer group together as well as the GU ASCO group put together, the FDA allowed us to conduct a single-arm study in CIS patients only, even though papillary patients were included because in CIS you start with cancer and you're treating cancer and then you have a CR which you can measure in a more reliable fashion than a papillary tumor, which is resected and depends a lot on the expertise of the resecting surgeon. I agree with Nick on that point about the papillary tumors. I just wanted to clear that up and then, Trinity, I'll hand the stage back to you.
Trinity Bivalacqua: Yeah, so thanks. Thanks, Nick, for those comments. They're all obviously important and excellent. I think I'd like to start off with the randomization a little bit and I know Ashish just went over why the FDA has allowed us to do that. In the United States, remember this was done in the United States. We don't have the ability to number one, to use hypothermic mitomycin C or any chemotherapeutic agent because the device is not FDA approved. We don't have that ability to do that. And I'm fully aware of all the data that's come out of Europe and the UK showing its utility. And I'll be honest with you, I actually believe in it. That's why I put looking to the future. I personally believe things like hypothermic chemotherapy, nanoparticle-based chemotherapy, that allows there to be better penetration through the bladder mucosa into the deeper wall of the bladder is I think the future. And I would acknowledge that.
As you know, because Professor Catto just published the paper in JCO showing that they were unable to randomize patients, at least upfront patients with T1G3 to either cystectomy versus BCG. It would be even harder to do that in the BCG unresponsive state because patients are looking for a bladder preserving approach. I don't think that that would be possible because as you know, we're all looking to preserve the bladder. That would be my only other comment there.
As it relates to endpoints, the endpoints are important for many reasons and the way I look at it is, is that we do need to look at the long term effects, three, five years to look at, as you pointed out, did those eight patients with muscle-invasive disease, are they dying? Are they dying sooner? Or have we missed our window of opportunity? And I acknowledge that information we need to learn more about as it relates to all of these agents. Same thing as it relates to pembro. And we as a group by going on and doing additional intravesical therapies or systemic agents, are we actually harming patients? And are they dying sooner from progression of metastatic disease? These are all endpoints that we need to include in future trials.
I also want to state that really, if you look at the data from this trial, Ad-interferon had the best efficacy in patients with papillary disease only. And I like to point out that 30% of the patients in the papillary disease only had T1G3. We're still looking at patients that had two or greater, more than 50% of the patients in the papillary disease had two or greater intravesical instillations of BCG, had high-grade T1, and we still are seeing response rates that are over 50% or around 50% at 12 months. I think that we as urologists are doing a better job of doing TURBTs to better stage. And I'd love to hear your comments on the utilization of MRI, for example. And I think that this agent at least has good activity in that disease itself. Your thoughts?
Nicholas James: Obviously, all sensible points. I'm intrigued by a number of things you said here. First is that normally urologists are desperate to take people's bladders out once there's a whiff of cancer in there. I spend most of my time trying to sway the urologists to do bladder preserving therapies instead of surgery. And here you are, propending a bladder preserving therapy so you don't do a cystectomy, which I do find not for you particularly, but I find a tad ironic, given the cystectomy bladder preservation debate a little bit to the right of this. I accept randomizing versus cystectomy is always going to be a really, really tough one. But I think de facto, most of these patients are likely to end up with cystectomies anyway. Roughly 15% of these people we know from historic series are going to die of muscle-invasive bladder cancer by five years out. We know this is a potentially lethal disease.
I think what you've shown is the equivalent of a sort of Phase II trial. In oncology terms, measuring response rates at three months or whatever the primary outcome was and the durability of response a year in a single-arm trial would be taken as proof of activity suitable for taking into a Phase III in a more robust way, rather than as a definitive thing in itself. Given that this is essentially an oncology trial, not a urology trial, albeit it's done by urologists, its endpoints don't really line up with how we generally license new oncology treatments as opposed to new surgical treatments. I think once you start looking at it from that point of view, I do find the FDA's approach to this a bit strange. And I think mostly my patients are interested in not dying of cancer rather than how many of them have had a response at three months. It's quite short term, given the length of the natural history of this thing.
Trinity Bivalacqua: Yeah, no, exactly. I will say that we are urologic oncologists, so I think we're oncologists as well. Your point is once again well taken, but remember we were under the guidance of what the FDA endpoints were in this disease state. I think we're going to start now that we have agents that have activity, we're going to start to be able to look at additional endpoints. And I will say the FDA is moving the bar with the design of new trials that are in BCG unresponsive disease. I think you're going to start to see these endpoints changing. We may see in the very near future, randomization to one of the FDA approved agents with a new study drug. I think we are moving the field forward. And I think this is an example of how we are hopefully making early advances and we'll continue to do that once we get more mature data.
Nicholas James: For me, the trial that would be very interesting to do, and I would suggest a multi-arm trial, although I'm sure it would be impossible to get set up because of the different companies involved, would be to do in the first-line setting. So, post TRBT, high-risk disease, stratified T1 versus CIS and randomize BCG or whatever you'd think that the standard of care versus everything you've got on the slide in front of me. That would be the way to resolve this. And you've clearly got an active agent. The question is, is it more active than BCG because BCG is frankly not a great treatment, is it? It works, but it's improvable on, I would say.
Trinity Bivalacqua: Yeah. Yeah. I think once again, we're seeing trials that are moving these agents into the BCG-naïve setting. I think we're going to have some actual information on that in hopefully 12 to 24 months because there's a number of trials that are looking at that.
Nicholas James: Because I think if you can show with a sort of a product that's manufacturable and put it in injectables out of a syringe versus a rather messy thing that carries quite significant risk, systemic BCG, very irritative problems, and a significant failure rate. There are multiple ways you can improve on BCG with a sort of more pharmaceutical grade product really, out of my field. And I think there's a reasonable chance any or all of these could do that.
Ashish Kamat: This has been a great discussion, guys. Couple of points that I do want to make a little bit since Nick and Trinity, you raise those. First off, going head-to-head against BCG is absolutely something we need to do. Except that being said, there is preclinical and other data that suggests that, for example, with pembrolizumab, [inaudible], priming with BCG, or some other agent might be acquired. Going head-to-head against BCG without adding BCG has been looked at and sort of discarded by a lot of groups. And there are studies where you're combining with BCG and again, full disclosure, nadofaragene was developed at MD Anderson now 15, 20 years ago. I remember working with mice in the lab when I was a fellow almost 20 years ago, but there are data and evidence to suggest that even with the adenovirus interferon, some level of priming with another agent in this case BCG, might be required. The head-to-head studies have not taken off for those reasons, mechanistic as well as logistics because you just require a huge number of patients, number one.
Number two, the data that everybody talks about when it comes to BCG not being tolerated is actually very old, more recent studies suggest that 90% of patients tolerate a full three-year course of BCG. It is very, very inexpensive, so inexpensive in fact that people have accused companies of not producing enough BCG because they don't make much of a profit. And if you look at more recent contemporary data from multiple centers, including Hopkins, the UK, and ours, the overall efficacy of BCG, including in T1 high-grade disease approaches 90% at five years with better resection, better optical enhanced technology to identify tumors, et cetera, et cetera. The bar is really high, but I agree with you. We need to do something to improve on BCG and whether it's combination or a single agent remains to be seen.
Gentlemen, I want to thank you. The three of us, we often talk for hours and hours on end, but our audience has a limited time to listen to us. I want to leave with an invitation to both of you, Trinity, 30 seconds, closing thoughts. And then Nick, I'll give you the honor of the final 30 seconds, closing thoughts on this matter.
Trinity Bivalacqua: Yeah, I think my only comment would be is that I'm looking forward to novel trial designs using Ad-interferon as a base agent, I do think it has activity. And I think that all this trial will do, is number one, it's going to help our patients. And it's going to help us learn about the resistance mechanisms that are occurring in the BCG unresponsive state. And it's going to allow us to do better as it relates to trial design, as well as improvement in efficacy. I think the future is bright.
Nicholas James: I'm going to have to agree with all of that. I think this is an exciting thing. It's a new therapeutic modality and it's clearly likely to be combinable with other things. And I think there's a lot of interesting opportunities that are going to come. I'm personally not entirely convinced by the endpoints if you're going to end up with a cystectomy median of nine months later, but nonetheless, it's clearly very exciting to have a new therapy and a lot of new therapies appearing in bladder cancer for the first time in many years, to be honest.
Ashish Kamat: Great. Thank you once again, Trinity and Nick. And in these times, all I can say is stay safe, stay well and see you in 2021.
Nicholas James: I very much hope so.
Trinity Bivalacqua: Thank you.
Nicholas James: Thank you very much and thank you very much for the invitation.


